The CQA Company

The CQA Company, acquired by Arithmos in November 2022, provides expert Quality Assurance advice to pharmaceutical, biotech and medical device companies globally.
Our GxP audits, quality management system development services and inspection readiness assessments are performed by highly qualified, specialist, regularly trained staff members, in line with current best practices and quality guidelines.
Our philosophy is one of teamwork, positivity, communication, responsibility and adding value. The CQA Company does not offer a theoretical approach to Quality Management and reviewing, instead all of our personnel have direct, hands-on experience in Clinical Quality Assurance and ICH GxP with the ability to provide a real-world insight into the role.
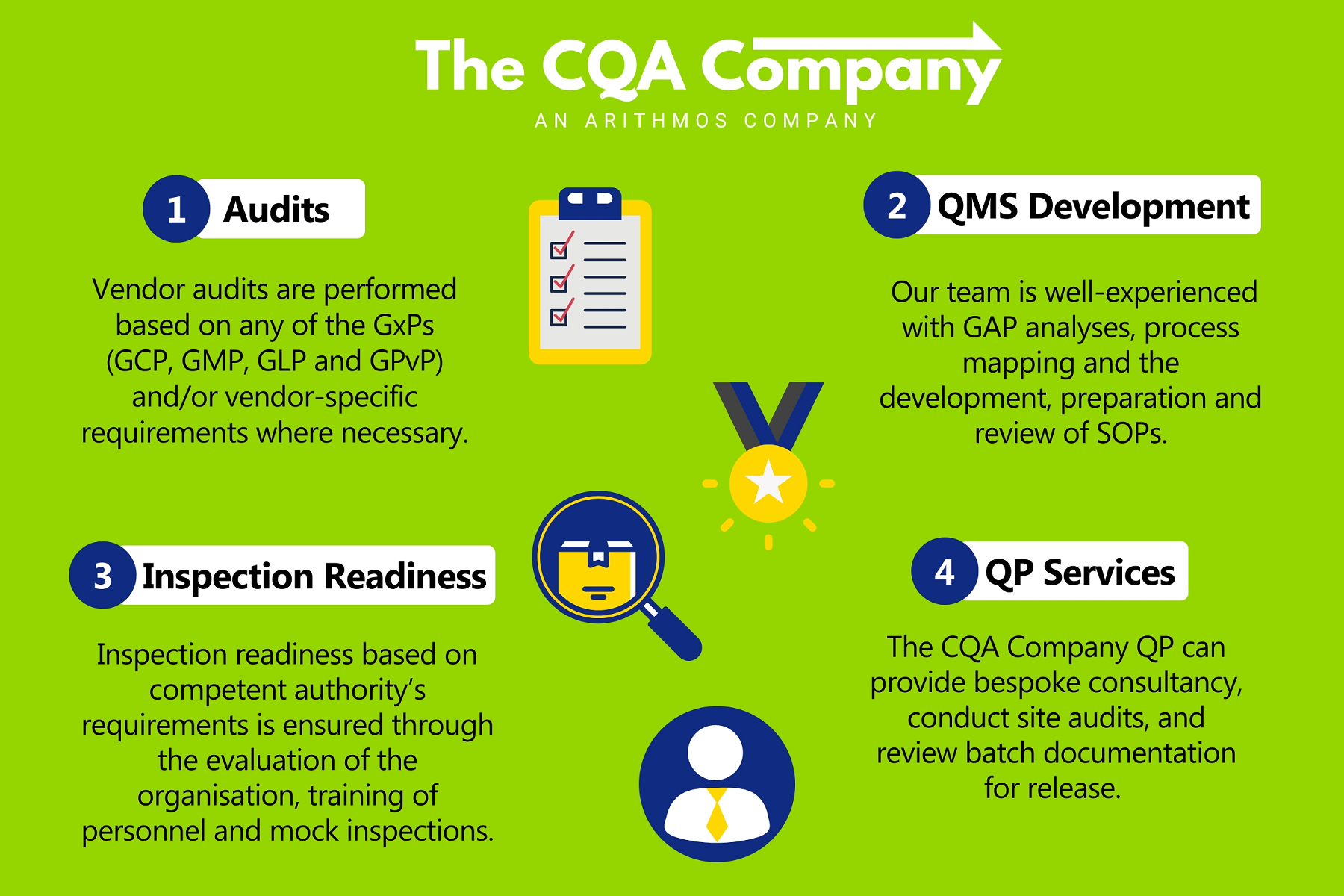
Our Services

GCP Audits
- Clinical Studies Phases I to IV
- Central Laboratories
- Clinical Laboratories
- Advance Products & Medical Devices
- Biotech environment
- Pharmacy
GMP Audits
- Raw Materials supplier
- Laboratory supplies (equipment/chemicals) & testing facilities
- Printed packaging material supplier & production facilities
- Product recall assessment
- Personnel training & hygiene
GPvP Audits
- PV/REMS Audits (EMA/FDA/MHRA) Sponsors & vendors
- EU – Audits of Risk Management Plans
- Clinical Site Audits
- REMS System audits
- PV inspection preparation/ hosting
GLP/GcLP Audits
- Facilities
- Laboratories (Equipment, Reagents & Materials)
- Study Performance
- Storage of records and reference material
- Data Reporting (LIMS)
Vendor Audits
- GCP, GMP, GLP, PV
- Clinical Research Organisations
- Phase I units
- Central Laboratories/Specialised Laboratory Testing Facilities
- Data Management
- Translation Companies
- IT and e-TMP
QMS Development
- Strategic Development
- QMS Assessment and legacy operations
- GAP studies
- SOP Development and preparation
- Process Mapping
- Risk Management
- Staff Training
- QP Services
Inspection Support
- EMA, FDA and MHRA experience
- Mock audits/inspections
- Pre-inspection training
- Mock inspection interviews
- Evaluation of company’s inspection readiness
- Inspection set-up
- On site inspection support
Our Knowledge

The CQA Company team is composed of highly experienced managers, auditors and coordinators covering all areas of clinical quality assurance.
Our auditors have a minimum of 5 years auditing experience and operate under the guidance of an EU and UK licensed QP.
The current Guideline for Good Clinical Practice (GCP), E6 (R2), was adopted by the EMA’s Committee for Medicinal Products for Human Use (CHMP) in December 2016 and came into effect in Europe in June 2017.
The CQA Company employs quality assurance specialists assigned to actively monitor updates to GxP guidelines and regulations, including draft updates (E6(R3)) published by ICH’s Expert Working Group, and this knowledge is passed on to our clients.
If you would like assistance with the incorporation of new guidelines and amendments into your Quality Management System, the CQA Company can supply the required Gap Analysis, Process Mapping, Risk Management and QMS development services to ensure your company maintains the highest levels of GCP compliance.





